Volcanic gases are volatile substances released from a volcano or volcanically active area. Volcanic gases are always involved in volcanic eruptions, but they are often given off by passively degassing (dormant) volcanoes as well. Volatile substances are chemical elements or compounds dissolved in magma that form a gas at relatively low pressure and temperature3.

Geothermal area in Iceland near Lake Myvatn is a beautiful place with unpleasant odor of hydrogen sulfide.
Composition of volcanic gases
It was once believed that each volcano has its own particular type of gas: sulfur dioxide at Etna, hydrogen chloride at Vesuvius, carbon dioxide at Puracé (Colombia), etc. In 1850s it was demonstrated by a French mineralogist Charles Sainte-Claire Deville to be untrue3. But each volcano nevertheless has its own character (average mixture of different gas species) which is a result of magma composition, tectonic setting, degassing depth, interactions with groundwater and hydrothermal systems, crustal composition, etc.
The most important constituent of volcanic gas is water vapor (forms about 90% of all the gases1) although the concentration is lower (about 60%) in gases coming directly from craters3. The next most important gas species is carbon dioxide (CO2) which makes up about 10…40% of gases. Sulfur compounds sulfur dioxide (SO2) and hydrogen sulfide (H2S) are also very important. Chemical elements Cl, F, and Br occur as acids (combined with hydrogen from water to form hydrochloric, hydrofluoric, and hydrobromic acid). They are also very common constituents although their composition is highly variable. Hydrogen (H2), carbon monoxide (CO), methane (CH4), sulfur (S2), nitrogen (N2), ammonia (NH3), oxygen (O2), and rare gases occur in smaller amounts. Numerous metals are also present (Pb, Zn, Cd, Hg, Cu, Bi, Na, K) at trace levels. They are mostly transported as chlorides and fluorides2.

Volcanic gases in Hawaii are rich in carbon dioxide and sulfur dioxide. Flank of Kilauea between Pu’u O’o and coast.

Degassing magma at the Halema’uma’u pit (collapse) crater within Kilauea summit caldera in Hawaii.

Icelandic landscape near the Krafla Volcano. Rocks in the foreground are covered with minerals (sulfur, gypsum) precipitated out from volcanic gases.
The sources of volcanic gases
There are three principal reservoirs which provide volatile components to form volcanic gas.
- The mantle
- The crust
- The atmosphere & hydrosphere
The material from the mantle dominates if the volcano itself is fed by a material from deep below. This is the case with hot spot volcanism under the oceanic crust. Good example is Hawaii. These volcanoes emit mixtures of gases that are relatively rich in sulfur and carbon compounds but, importantly, contain little water. This is the very reason that explains why Hawaii and similar island volcanoes far away from the continental margins are not very explosive – they do not emit much water vapor. It is the buildup of gases within the volcanic edifice that results in explosive behavior.
Volcanic gases of the volcanoes on the continental margins (the Ring of Fire volcanoes surrounding the Pacific Basin, for example) are compositionally more versatile and variable because they also incorporate the crustal material which may have a highly variable composition. These volcanoes are generally explosive in nature because their magma is thick (more siliceous composition hinders the release of volatile substances) and it also contains lots of water which expands more than 1000 times when it goes from dissolved state (within magma) into separate gaseous phase.

Mudpot is a type of fumarole. Acidic volcanic gases corrode the surrounding rocks, turning them into watery slurry which appears to be boiling because of bubbling. This mudpot is in Iceland near Krafla volcano.
Where does that water come from? There are two main sources. This type of volcanism is associated with subduction zones. Subducted slabs of oceanic crust carry hydrated minerals beneath the continental crust where they start releasing water because of immense pressure. This water additionally helps to melt the rocks which start to rise as liquid magma and will eventually create a chain of volcanoes on the continental margin (like The Cascades, for instance). This water was originally part of the hydrosphere-atmosphere system but because it went through the subduction process it is no longer considered to be a part of it. Another major source of water in volcanic gases is meteoric water. This is basically rainwater either directly or indirectly through water bodies containing rainwater. The hydrosphere-atmosphere system does not only provide water but also substances that are dissolved in water (oxygen and other atmospheric gases). The particular mixture of volcanic gases is therefore highly dependent on local conditions.
It is also worth mentioning that volcanic gas (or part of it) does not need to be a result of magma degassing. It may be just groundwater heated by nearby volcanism. Volcanic gases may be released from summit craters but also from cracks on the flanks (fumaroles) or they may be released diffusively through the ground (especially nonreactive species like CO2 and He3).

Sulphur Springs, Saint Lucia. Saint Lucia is a volcanic island located on top of a subduction zone.
The influence of volcanic gases on climate and environment
Scientists are obviously studying volcanic gases to gain an insight into the inner workings of a volcano to be better prepared for future eruptions. But it is also very interesting to study the gases because they have immensely influenced the composition of the Earth’s atmosphere (and also hydrosphere) in the past and continue to do so in the future as well.
The Earth’s atmosphere and hydrosphere influence the composition of volcanic gases to great extent but it is interesting to note that both the atmosphere and hydrosphere are the results of magma degassing. All of the major constituents (nitrogen, carbon dioxide, water) of these geospheres are volcanic in origin (notable exception is oxygen). The primordial Earth was probably entirely molten. As soon as it cooled the water started to accumulate in lower areas as rainwater condensed from volcanic clouds of steam. Primordial atmosphere was also very rich in carbon dioxide. Current level 0.04% may seem much to us because only a few decades ago it was 0.035%. It is a major and very rapid change most likely caused by human activity. But the Earth has seen times when the level of CO2 has been many tens of times higher. Even during relatively recent history (recent when compared with the entire 4.5 billion years of Earth’s history) when dinosaurs ruled the Earth there were at least five times as much CO2 in the air. Consequently the average air temperature was also significantly higher (about 22 °C) because of more intense greenhouse effect.

Sulfur precipitated from volcanic gases in Iceland, Krafla Volcano. Sulfur compounds (especially sulfur dioxide) have a potential to severely affect the climate and environment.
Volcanic gases are therefore highly influential variables modifying the Earth’s climate but the way they influence it is by no means simple and straightforward. Carbon dioxide and many other volatile compounds released from volcanoes (like water vapor and sulfur dioxide) are potent greenhouse gases but some substances have the opposite cooling effect. SO2 especially is noteworthy for both cooling and warming role. It warms the atmosphere as a greenhouse gas but it also forms sulfuric acid aerosols which can stay in the stratosphere for several years and have a clearly cooling effect as a haze that backscatters incoming solar radiation.
The vast majority of volcanic eruptions have very limited and local effect on climate and environment. This is because (1) the amount of gases released is small, (2) the eruption does not last long enough, (3) the eruption cloud does not reach high enough to penetrate into the stratosphere. The latter is very important because tropospheric aerosols will be washed out within a week. The duration is also very important because in order to have a long-lasting effect on the environment the ocean-atmosphere climate system must come to equilibrium with volcanic perturbations. That is not going to happen if the eruption lasts only few months even if it is relatively powerful.

Geothermal area near Strokkur (the most famous geyser in Iceland) in Iceland.
The most severe effect on climate comes from very large-scale and long-lasting basaltic eruptions which are known as flood basalt events. Why does the eruption need to be basaltic? Does not common silicic eruption contain more volcanic gases? Yes, it is true but these gases that cause powerful silicic eruptions are largely composed of water vapor which will not wreak havoc on the Earth’s climate and environment. Basaltic eruptions on the other hand contain more sulfur dioxide which has much more potential to cause long term environmental harm. Flood basalt events are so influential because of powerfulness, long duration, and lots of sulfur.
Nowadays scientists are more and more linking flood basalt formations with major extinction events in the Earth’s history. The extinction of dinosaurs 65 million years ago has long been associated with the impact event on the Yucatán peninsula which undoubtedly had a negative effect on the environment. On the other hand, it is unlikely to be the only culprit because it was instantaneous event which had short-lived effects. It seems more likely that we need hundreds if not thousands of years to constantly alter the ocean-atmosphere chemistry to cause mass extinctions. Further credit to this hypothesis comes from the fact that while the End-Cretaceous extinction event which ended the rule of dinosaurs is associated with the Deccan flood basalt event. Another and much more severe extinction event 250 million years ago is also coeval with a major flood basalt formation in Siberia.
While volcanoes may be responsible for large extinction events they also probably have saved life on Earth at least once. About 650 million years ago. This time in the Earth’s history is known as the Snowball Earth event. Most of the Earth was covered with ice which significantly increased the reflectiveness (albedo) of the Earth which helped to cool the Earth even further. It should be a situation with no return to normal conditions. However, somehow it happened. Probably because of intensified volcanic activity which pumped large amount of greenhouse gases into the atmosphere.
Volcanoes and volcanic gases in particular have a significant and complicated role in modifying the climate and atmosphere. We know that current background level of volcanism is not significantly contributing to global warming or altering the Earth’s environment. But the Earth’s history has demonstrated clearly that they have a power to do so.

Fumarole on Kilauea Iki lava lake in Hawaii.
Hazards associated with volcanic gases
Volcanic gases seem to be a relatively minor hazard if we count the casualties while keeping the fatalities caused by the pyroclastic density currents (PDC) aside. PDCs are very deadly but this phenomenon is usually treated separately because it is not only a fiery gas cloud but also a mixture of variously sized rocky material. Although the overall number of deaths caused by volcanic gases is relatively modest, asphyxiation is definitely not a pleasant way to die. What are the dangers then and what can be done to avoid them?
Most of the common constituents of volcanic gases are hazardous to humans and other living forms in elevated concentrations. The main culprits causing deaths or health problems are carbon dioxide, sulfur dioxide, hydrogen sulfide, hydrochloric acid, hydrofluoric acid, and sulfuric acid.

Volcanic gases rising from the summit caldera of Kilauea Volcano in Hawaii.
Carbon dioxide
Carbon dioxide is harmless in small quantities (0.04% of air is CO2) but can cause almost instant coma if concentrated enough. CO2 is odorless and colorless and denser than air. It means that the danger is difficult to detect but we have to make sure that in volcanically active areas we will think twice before going to explore any kind of holes, craters or depressions because they may be full of this gas. Carbon dioxide has caused large number of casualties associated with volcanic gases. Especially famous is the Nyos Lake event which killed 1700 people in Cameroon. I am not going into details because this event is already well described elsewhere and second because it is a remarkable but by no means widespread event. There are only two instances (Nyos and Nearby Monoun Lake events in 1986 and 1984, respectively) when such a rapid large scale release of carbon dioxide from lakes are documented4.
Carbon dioxide is also lethal to vegetation although in smaller quantities all green plants need it for photosynthesis. Even large trees can be killed if there is so much CO2 in the soil that the roots of trees can not absorb oxygen anymore. The most famous such area is in the Inyo National Forest in California. My personal funny and sad story associated with this place is that although I consider myself a hobby photographer of everything geological I apparently failed to take a picture of this place although I saw these dried trees there with my own eyes. It happens, I suppose, especially if you are stupid enough to think that dead trees have nothing to do with geology.
Sulfur dioxide
Sulfur dioxide has a distinct sharply penetrating odor which is detectable already in concentrations as low as 1 ppm (one part per million). I remember one breath on the flanks of Kilauea near flowing lava which was so sharply irritating to my nose that made me instantly turn and run. It was the smell of SO2 in concentration well above 1 ppm. Perhaps you have felt it also. When lighting a match too close to your nose you may feel a burning and unpleasant sensation which is caused by the same sulfur compound. Sulfur dioxide reacts with water in the air to form sulfuric acid which precipitates as acid rain and also backscatters radiation from the Sun, thereby lowering the average temperature.

Volcanic gases are often rich in sulfur, especially lavas of mafic composition. Bright yellow mineral is elemental sulfur. Sulfurous fumarole is known as solfatara. Sulphur Banks, Hawaii.
Hydrogen sulfide
Hydrogen sulfide is another toxic gas which also has distinct and rather unpleasant smell often associated with rotten eggs. That, I am afraid, is not too helpful for most of the people because rotten eggs seem to be a hard to find curiosity nowadays. Anyway, this sulfur compound and its odor are easily noticeable and very common in volcanically active areas. That gas is also heavier than air and tends to concentrate in lower areas. In 1971 six downhill skiers in Japan died almost instantly when passing through a depression filled with it4. Many other volcanic gas related fatalities are also directly associated with this sulfur compound.
HCl and HF
Fumes coming from volcanic areas are acidic not only because of sulfuric acid but also because of hydrogen chloride and hydrogen fluoride. The first one is the principal constituent of gastric acid and carried also in a small bottle by every serious field geologist because it makes an easy test for carbonate rocks (to determine whether it is limestone or dolostone). HCl is not really very serious threat to our lives (although it has caused fatalities, of course) but it contributes to acid rain, does cause breathing problems and is irritating to the eyes. In Hawaii, lava haze (or laze) is forming when molten lava flowing to the sea initiates a chemical reaction between salt ions dissolved in seawater and water which results in hydrochloric acid. Hydrofluoric acid (HF) is also used by geologists but this time in laboratories in carefully controlled conditions because this substance is really nasty stuff. It is extremely irritating to the skin and burns heal slowly. Contact with the eyes will cause serious burns and blindness if not removed immediately with running water.
http://picasaweb.google.com/107509377372007544953/Rocks#5877841693091516370
Lava laze (lava + haze) in Hawaii. This cloud of smoke is composed of water vapor but it also contains lots of hydrochloric acid. It rises from the place where lava flows onto the sea from a lava tunnel.
http://picasaweb.google.com/107509377372007544953/Rocks#5877841703074302994
Hydrochloric acid forms as seawater reacts with salt (sodium chloride) dissolved within it.
So what can we do to avoid dangers associated with volcanic gases. It is really easy. Stay away from volcanoes and nothing bad happens. However, if this is not what you wanted to hear then I recommend to definitely avoid lower areas where dense gases can accumulate and also try to pay attention to the wind direction. Try to stay windward (upwind) from the sources of volcanic gases and if you really have to go to really dangerous places then you hopefully already know what you are doing because you are a professional volcanologist with a good gas mask and protective clothing.
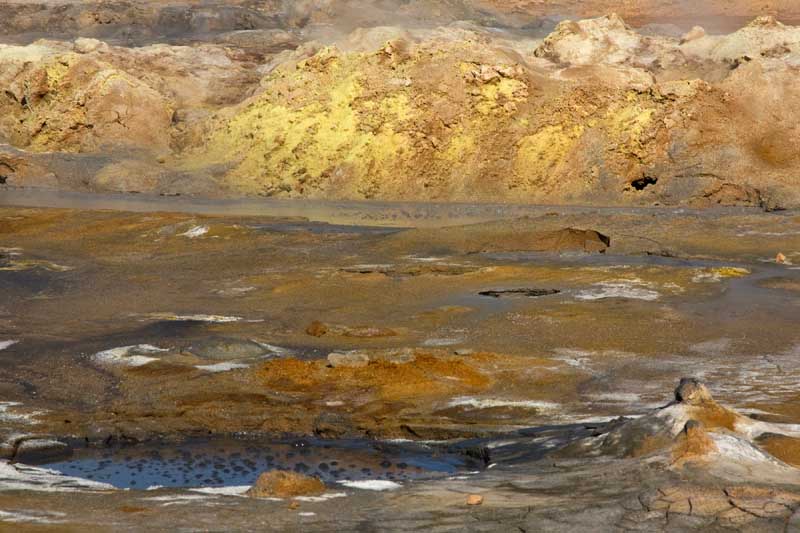
Geothermal area in Iceland near Viti Maar with mudpots and yellow sulfur.

Muddy fumarole (mudpot) in Iceland.

Another mudpot in Iceland.
References
1. Jackson, J. A. (1997). Glossary of Geology, 4th Edition. American Geological Institute.
2. Gerlach, T. M. (1999). Volcanic Gases. In: Encyclopedia of Geochemistry (Encyclopedia of Earth Sciences Series) (Ed. Marshall, Clare P. & Fairbridge, Rhodes W.). Springer. 656-657.
3. Delmelle, Pierre & Stix, John (1999). Volcanic Gases. In: Encyclopedia of Volcanoes (Ed. Sigurdsson, H.). Academic Press. 803-815.
4. Williams-Jones, Glyn & Rymer, Hazel (1999). Hazards of Volcanic Gases. In: Encyclopedia of Volcanoes (Ed. Sigurdsson, H.). Academic Press. 997-1004.
Leave a Reply